Key Takeaways
|
GLP-1 medications, such as semaglutide (Ozempic®, Wegovy®) and tirzepatide (Mounjaro®, Zepbound®), have transformed the clinical approach to weight management. In fact, an estimated 1 in 8 adults in the U.S. have used a GLP-1 medication in recent years.[1]
Yet, if you or someone you know has taken one, you might have noticed that not everyone has the same experience on these drugs. While GLP-1 medications are commonly prescribed to help control blood sugar and to assist in weight loss, there is substantial variation in how well they work for different people. Some individuals lose as little as 5% of their body weight, whereas others lose more than 20%.[2][3] Similarly, while gastrointestinal side effects such as nausea and vomiting are common, some people experience moderate to severe forms, whereas others report no nausea at all.[4]
While lifestyle characteristics like diet patterns and exercise may certainly explain some of these differences, potential genetic causes of this variation have been unclear, until today. Scientists at the 23andMe Research Institute published a study in Nature that has identified genetic variants associated with weight loss and side effects among those who have taken GLP-1 medications.
The Mystery of Medication Response
When people begin a GLP-1 journey, they may start with uncertainty about efficacy and possible side effects. While the market is crowded with weight loss support and medications, few resources offer personalized insight into the simple question: “What can I expect?”.
To find answers, we turned to our incredible community of 23andMe research participants. Over 27,000 consented participants made this research possible by sharing their experiences with GLP-1 medications through an ongoing research survey. Using 23andMe’s unique crowdsourced approach to research, we were able to conduct the first large-scale genome-wide association study (GWAS) just seven months after starting the study, a very rapid rate for a study of this kind. In simple terms, a GWAS is like scanning a massive library of DNA to find specific “spelling differences” (genetic variants) that are more common in people who share a specific trait. In this case, scientists wanted to understand if there were genetic differences that were associated with how participants’ bodies responded to GLP-1 medications.
Discovering the Genetic Links
23andMe Research Institute scientists uncovered fascinating genetic associations for response to GLP-1 medications, including a person’s potential for weight loss and their risk of experiencing nausea or vomiting.
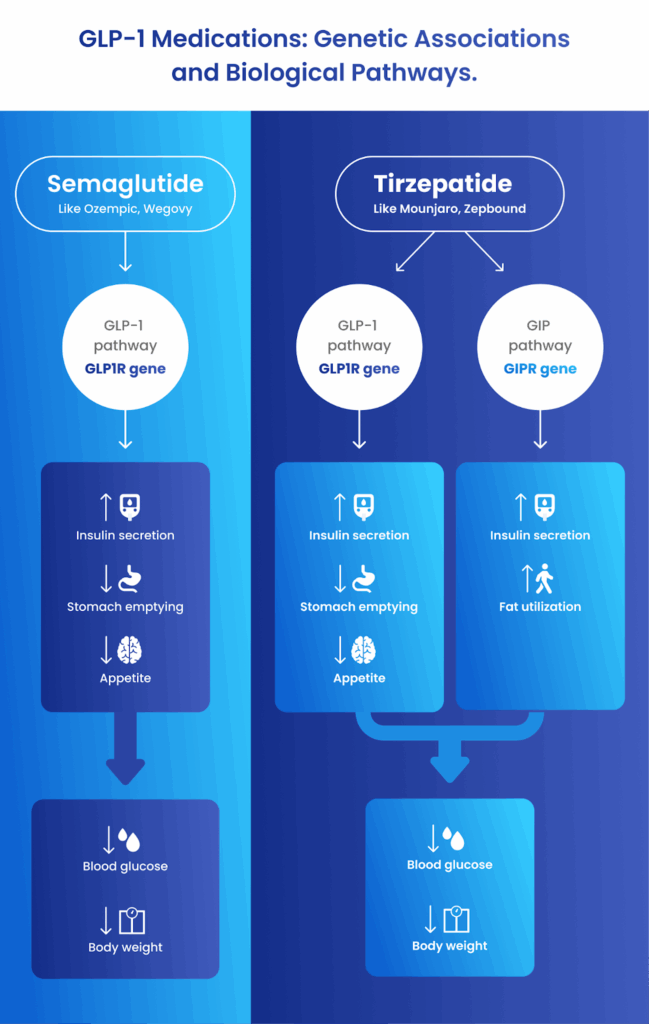
First, the study identified a genetic variant that is associated with both weight loss and nausea for the two major classes of GLP1 medications: semaglutide and tirzepatide. This variant is located in the GLP1R gene, which codes for the receptor protein that is the target of GLP-1 medications.
Second, the study identified another genetic variant that is associated with side-effects, specifically nausea and vomiting, while using tirzepatide (Mounjaro® or Zepbound®). You might wonder why this second variant only affects tirzepatide users. It comes down to how the drugs are designed: while semaglutide acts only on GLP-1 receptors, tirzepatide is a dual-target drug that also acts on GIP receptors (GIP, or gastric inhibitory polypeptide, is another hormone like GLP-1 that helps regulate insulin and blood sugar). Our team discovered that this second variant is located in the GIPR gene, which explains why it impacts tirzepatide response but not semaglutide.
Describing the impact of the study, Dr. Adam Auton, Vice President of Human Genetics at the 23andMe Research Institute said, “Identifying these variants in the GLP1R and GIPR genes provides an important new insight into why these medications impact people in distinct ways. It’s a great example of how our large-scale, participant-engaged research program can clarify the biological mechanisms behind drug response, and moves us closer to a future where ‘precision medicine’ isn’t just an aspiration.”
Translating Emerging Science into Personalized Insights
Discovering these variants is a massive step forward, but our ultimate goal is to meet the need for more personalized insight into GLP-1 medication responses.
That is why a new report and interactive tool related to GLP-1 medications is now available exclusively through the 23andMe+ Total Health™ service. This interactive tool combines an individual’s genetic results for these two variants with demographics and medical history to estimate both weight loss and nausea likelihood based on data from 23andMe research participants. All these factors combined can result in significant variability in the likelihood of weight loss or side effects. In fact, the estimates for weight loss can vary between -6% to -20% of baseline weight after one year. Likewise, the estimated risk of nausea and vomiting can range between 5% to 78% risk for experiencing these side effects.
What You Need to Know About the New Report
The science of genetics is always evolving and the 23andMe Research Institute strives to be on the forefront of genetics research, powered by the participation of our members. Our mission is also to help people benefit from genetics research, which is why we are thrilled to provide this new report to our Total Health members. This report is designed to be reviewed in consultation with a clinician and is intended for use in a supervised clinical context, which is why it is exclusively available through the physician-supervised Total Health service.
The shift toward personalized care enabled by this report is echoed by Dr. Noura Abul-Husn, Chief Medical Officer of the 23andMe Research Institute, who views the data as a catalyst for better patient-doctor relationships: “For healthcare professionals, the true value of this data lies in its potential to transform weight loss discussions from a trial-and-error approach into data-driven, shared decision-making with patients. Integrating genetic markers, such as those we’ve identified in the GLP1R and GIPR genes, with clinical factors provides a clearer picture of how a patient’s unique biology may interact with a specific therapy, allowing both patients and their doctors to proactively manage expectations and tailor care with greater precision from day one.”
A Sincere Thank You to Our Participants
The GLP-1 Study is ongoing, and our survey is designed to allow participants to share their journey over time so we can continue to investigate how these medications are associated with genetics and health. Discoveries like this simply would not exist without our research participants. Current 23andMe members can choose to contribute answers and to fuel new discoveries.
Participate in the next discovery
References
1. Montero, A., Sparks, G., Presiado, M. & Hamel, L. KFF Health Tracking Poll May 2024: The Public’s Use and Views of GLP-1 Drugs | KFF.
2. Wilding, J. P. H. et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. New Engl J Med 384, 989–1002 (2021). https://pubmed.ncbi.nlm.nih.gov/33567185/
3. Ryan, D. H. et al. Long-term weight loss effects of semaglutide in obesity without diabetes in the SELECT trial. Nat Med 30, 2049–2057 (2024). https://pubmed.ncbi.nlm.nih.gov/38740993/
4. Joy, Bethany, et al. Exploring the rates of Gastrointestinal adverse effects among five GLP-1 receptor agonists: A systematic review and Meta-Analysis of randomized controlled trials. Endocrine 91(1), 23 (2026). https://pubmed.ncbi.nlm.nih.gov/41489846/




